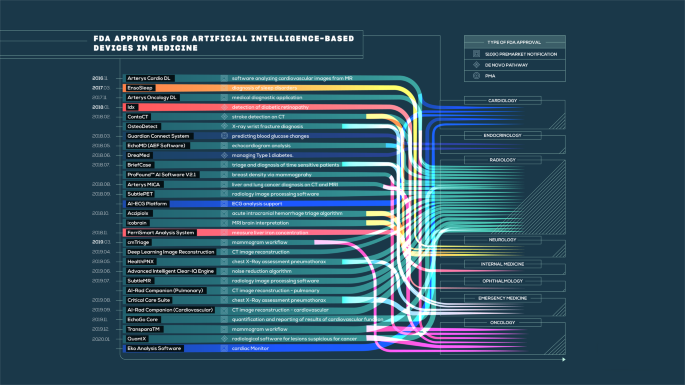
The state of artificial intelligence-based FDA-approved medical devices and algorithms: an online database | npj Digital Medicine

U.S. FDA on Twitter: "They're not the prettiest, BUT they're still safe, nutritious, and delicious, so don't count the imperfect vegetables out 🥦🥕🌽. https://t.co/Z0xw5a0LDI #NationalEatYourVegetablesDay… https ://t.co/tfar1on2wx"
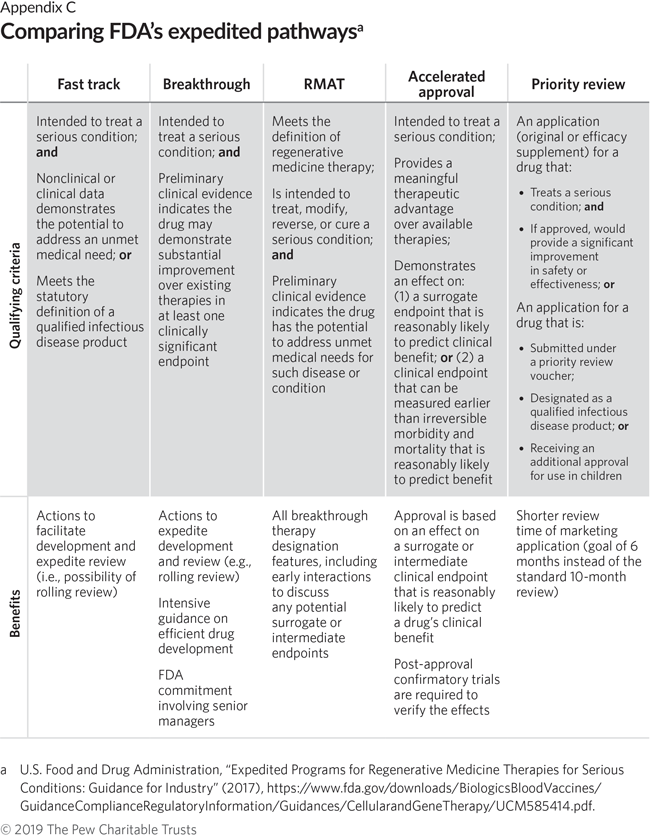
FDAs Framework for Regulating Regenerative Medicine Will Improve Oversight | The Pew Charitable Trusts

U.S. Food and Drug Administration - Did you know? Generic drugs saved the U.S. healthcare system nearly $2.2 trillion from 2009 to 2019! Generic drugs work in the same way and provide
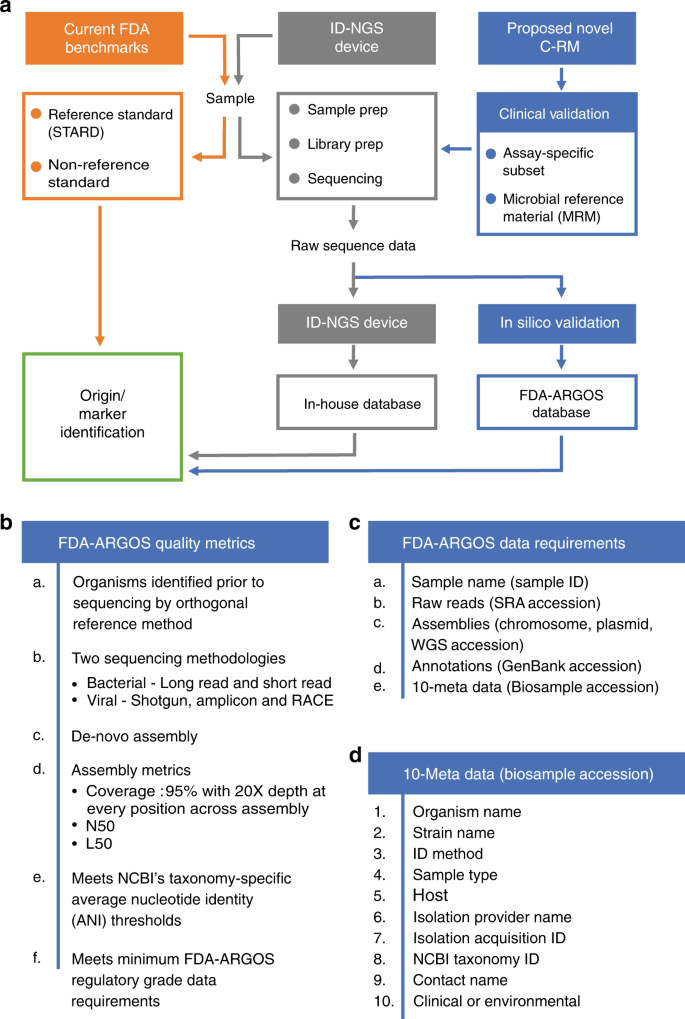
FDA-ARGOS is a database with public quality-controlled reference genomes for diagnostic use and regulatory science | Nature Communications